Six underdiscussed obstacles for new medical technologies
Obstacles for entrepreneurs and innovators to know and avoid
I. An invisible engine of progress
Pop quiz: what does treating these conditions have in common?
Take a moment and try to crack this one. Consider how these conditions are treated, on the most fundamental level.
Have you figured it out?
There are several answers, but the simplest one is that for all of them, the best possible treatment requires modern medical devices. Medical devices are fundamental for modern healthcare, and for the health outcomes that we take for granted. Take away the tools, and healthcare can't treat. However, we often forget this.1
I've met many brilliant people who have spent decades of their lives on improving healthcare by creating new medical technologies. However, as several have experienced, even if a new medical device can reduce costs or improve quality of care, providers may still not be interested in adopting it, and commercialising the technology can be challenging.
I’ve met many aspiring entrepreneurs who are passionate about improving healthcare. However, some don’t understand how healthcare works and why providers aren’t interested in using new technology. Throughout the years, I’ve noticed that some obstacles recur and are under-discussed, so I decided to summarize them below.
There are several obstacles which are commonly discussed in medtech books and blogs.2 I will not discuss those. Here I’ll focus on six important obstacles that are rarely discussed.
Grossly simplified, a new medical device can improve healthcare in one of two ways3:
Reduce costs
Improve quality/outcomes
However, even if an innovator knows that their technology can do one or both of the above, they can still encounter obstacles. For example…
II. It reduces costs, but…
It’s not enough for new medical technologies to reduce costs. Technologies will face challenges to adoption even if they reduce healthcare costs but...
2.1 Not for the stakeholders who decide to acquire and implement it
I once encountered a remote patient monitoring (RPM) system which heart failure patients could use at home to improve their medication and reduce deteriorations, emergency admissions and hospital length of stay. In contrast to many other RPM solutions, this one had several high-quality studies showing that it worked and could save lives and reduce costs.4 Despite that, it never got implemented on a larger scale.
Why? One reason was that the cost for the system had to be paid by the primary care budget, but the savings occurred in the hospital budget. The primary care decision maker had no financial incentive to purchase it - even though the solution would improve outcomes, save money for society and save time for patients and providers.
I’ve seen this in a depressingly large number of other cases. As healthcare specializes, most systems inevitably drift towards having a greater number of separate units and budgets, which results in this type of budget misalignment becoming more common.
It’s a pity that budget structures (a social construct and management mechanism) can block the adoption of new vital medical devices. However, being aware of this early on, and planning ahead can help innovators avoid that pitfall.
2.2 Not in this budget cycle
Ask any healthcare manager anywhere and she will tell you two truths: there is always a budget, and you always have to meet it. Even if a long-term investment could improve health outcomes significantly, that will be much more difficult to implement than an investment which yields smaller improvements but in a shorter timespan. The longer away the savings occur, the more certain that provider must be that the savings will happen and that they will be significant.
Similarly, the larger the investment and the further away the savings will occur, the broader this conviction has to be held within the organization, in order for there to be enough support for it to happen. This conviction will require scientific evidence and other data supporting that the technology can generate the desired effects.
Like all other obstacles, this isn’t binary, but rather a spectrum. Healthcare does in fact do many long-term investments. Building new hospitals might be the clearest and most generic example (requiring a lot of analysis and broad political support). For devices, the implementation of radiosurgery, like the Leksell Gamma Knife (with hundreds of publications) makes the specific point. Ensuring early proofs of value, or having a plan on how to create the relevant data can assist, but neither negate this challenge.
Innovators often need to collect different types of data at different stages of their journey. This is especially clear for the still nascent field of digital therapeutics, as illustrated in this graph.
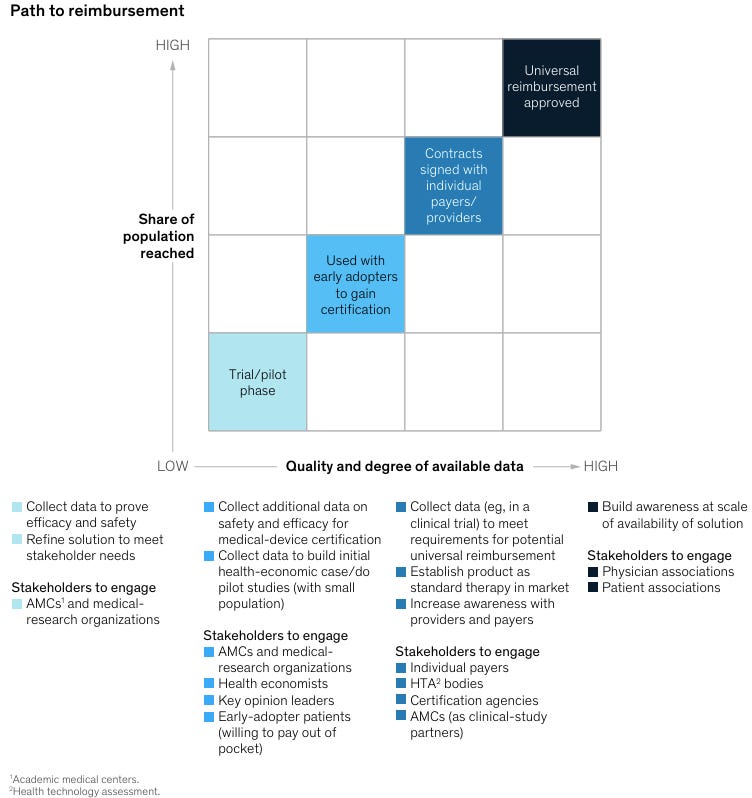
2.3 Not in a sufficiently large amount for each decision-maker
Providers have a limited amount of time and mental bandwidth. There isn’t time to understand, assess and spend time on new technologies which can’t generate significant cost reductions. Would you spend time on something that could reduce your personal monthly costs by 0.06%? Well, neither would (or should) a healthcare manager. New innovations need to have a sizeable financial impact.
Since healthcare is very big (~9% of GDP in the OECD), improving any one little thing across all of healthcare can be worth many billions of dollars. “How much money could this save across all of healthcare?” is the wrong question to ask to understand whether a provider will adopt a technology. The right question is “What share of the total costs for in this decision-maker’s budget can this reduce?”.
Even if something could save billions of dollars across all of healthcare, adoption will be slow and challenging if it only saves a small percentage of cost for each individual decision-maker.
On that note, a common misconception is that if a new technology can save time for clinicians, it can therefore also reduce costs for providers. This is rarely true.

Many technologies aim to reduce costs by saving time for clinicians. Staff costs are the largest variable cost for all healthcare providers. If you want to have a big impact, you need to either save time for clinicians or reduce costs for equipment.
However, time savings most often don’t actually lead to cost reductions. Providers won’t suddenly start firing staff or reducing staff hours. You don’t suddenly reduce the cost of an operating room if you can finish an operation 5 minutes earlier (you can’t suddenly reduce staff hours or reduce the cost of the equipment).5 Instead, the time that’s saved is often recouped by clinicians (who then have time to go to the bathroom) or by the provision of more care (i.e. increasing production or revenues for the provider).6
This might seem like a semantic difference, but plays a major role for the healthcare provider. Even if a technology does increase efficiency, this might not translate to cost reductions, and they therefore need to figure out how to fit this new cost into their budget. The better innovators understand this, the more realistic and relevant business cases they'll be able to create.
Now, beyond obstacles in reducing costs, there are also similar under-discussed problems in improving outcomes and quality of care.
III. It improves quality, but…
New medical technologies will face challenges even if they improve quality of care or performance, but…
3.1 Improvement can’t be measured
First, there are many different types of outcomes and quality to improve in healthcare. Each medical condition (e.g. heart failure) has many different outcomes which can be worth assessing.7 Typically, most providers can neither measure nor follow all of these outcomes.
Second, quality and outcomes can be measured in different ways. For example, an important thing like patient experience can be measured in different ways, which capture different aspects of what the patient experiences.8 Depending on the scale currently in use at a certain provider, some aspects of the experience might be difficult to capture.

Third, some outcomes can’t be measured. When a patient fills in a survey about their quality of life, does the aggregate number really capture all the nuances and richness in the lived experience? No, several aspects are lost. Improvements which are deeply subjective, or difficult to express and quantify, might not be measurable in a meaningful way.
Or, stated bluntly by Loadsman et al 2019:
Given the multidimensional and very individual nature of the pain experience, the concept of quantifying any alteration in pain as a percentage is arguably absurd
Adoption of a new technology will be much more difficult if an innovation improves something that isn’t currently or can’t be measured. Healthcare providers don’t like additional administration (there is a reason why the essay Why healthcare has so much administration has been read by many thousands of readers), and having to both implement a new technology (which will require new ways of working) and also setting up new measurements of a new outcome will be a big issue for many providers. Ensuring that one’s innovation’s improvements can and are being measured can greatly facilitate adoption.
3.2 Improvement isn’t prioritized
As there are many different outcomes for any given condition, any provider must prioritize which outcomes to improve and to what level. Even if a technology can greatly improve a certain outcome, let’s say outcome X from 70% to 85%, a certain provider might still want to spend more time on raising another outcome Y from 30% to 60%.
In other words, it’s necessary but not sufficient to find a large unmet need - entrepreneurs and innovators must understand which improvements clinicians deeply care about and prioritize over other improvements.
3.3 Improvement isn’t reimbursed
Reimbursement has different roles in different healthcare systems, so this depends on which country you’re looking at.
Lack of reimbursement: In general, if a technology clearly reduces costs or improves quality, it doesn’t always need specific reimbursement. However, reimbursement is key for certain technologies. My favorite example is digital therapeutics (DTx), which is software for treating or preventing medical conditions.9
Digital therapeutics have a large potential as they lack of side effects, need to prove efficacy prior to approval, are easy to distribute and can be scaled with low marginal cost. However, adoption in Europe has been slow, in part due to that they aren’t reimbursed in most of Europe, and when they are reimbursed it’s often confusing10. The US is better, where reimbursement now is possible through specific CPT codes.
Another medtech example: pacemakers are implanted into patients to treat certain conditions when the heart doesn’t beat as it should. Patients with pacemakers visit their cardiology clinic every year so that the cardiologist can export data from the pacemaker device and see how the patient’s heart has been beating. In 2009, the first pacemaker with the ability to export this data wirelessly from patients’ homes was used. This little yet brilliant innovation meant that you could, in many cases, remove a physical visit to a clinic and instead monitor the patient from afar.
When I discussed this with a leading cardiologist in 2014, he explained that the pacemakers they implanted had that ability, but that they had to continue with physical visits as there was no reimbursement for monitoring the distance from afar. Big medtech giants can afford to innovate and wait for several years until reimbursement catches up. However, for e.g. a small startup, this lack of reimbursement can be fatal.
Reimbursed too low, so that providers lose money: Even if there is reimbursement, a too low level can impede adoption. For instance, there are digital therapeutics which treat depression and can replace visits to a clinic with interactions with an app. However, if clinician visits are reimbursed more than the use of digital therapeutics, implementing the depression treatment app would reduce the clinic’s budget, something few managers would choose.
This problem is unfortunately even clearer for novel medical devices which enable intrahospital operations to become day surgeries. Intrahospital surgeries cost more than day surgeries and are therefore reimbursed with more money. Even if a novel technology could enable more day surgeries, with lower costs and complication rates, this would also reduce the total reimbursement to the clinic, which few managers would choose.
No one said it would be easy11 to develop new medical devices. Beyond these six obstacles, there are many other challenges to overcome and intricacies to navigate. But few things worth having come easy. And hopefully these concepts will increase the chance that some innovation out there will end up reaching and helping patients.
IV. Emmetropes
New medical technology is central for progress in healthcare
Adoption of novel medical technology requires more than solid evidence and proof of benefits for patients or providers
If researchers, inventors and entrepreneurs design technologies to better adress the following challenges, more innovations will reach and help patients
This becomes abundantly clear in a prehospital setting, where clinicians are limited in what you can diagnose and treat - mainly because you don’t have all the devices, machines and medicines that you have in a hospital
For example here. These aspects are all important: make sure you’re solving an important need, figure out way to generate the needed scientific evidence, ensure a feasible and effective regulatory plan, engage key opinion leaders for relevant markets (i.e. US) etc.
Astute readers will be aware of the third category: increasing revenues. In general, this is either due increased efficiency (i.e. delivering more of the same care) which is a type of cost reduction, or by allowing a healthcare provider to offer new services. Being able to expand offerings is important for an individual provider, but less relevant for innovators trying to implement new technologies, which is why I won’t cover that here
Technologies that prevent deteriorations may either prevent or postpone them. In the former case this can generate a cost saving, in the latter one shifts costs to next year's budget. This technology seemed to primarily prevent deteriorations, and actually generate a cost saving for the healthcare system.
Some nuance: you can reduce OR costs significantly if you reduce operation times significantly, consistently, and predictably.
This survey illustrates my point. A couple of years ago I discussed this with a leader of a leading management consultancy firm’s healthcare practice. Their experience, from a decade across several continents, was that the first half hour that is saved is reclaimed by the clinician, and that you only get disposable time after that.
For example, for congestive heart failure, one could consider following ICHOM’s eight categories of outcome measures: hospital visits, treatment side effects, complications of treatment, vital status, symptom control, psychosocial health, independence and activities of daily living
This article lists four different scales for assessing patient experience of hospital care. If you look in Table 1 you’ll see that the scales have different lengths and assess different aspects. For example, unless you’re using the PEQ scale, capturing improvements in patients’ emotions









Thank you, Jonathan !
Insightful! This is all very similar to the challenges of selling software to Enterprise companies in general. Often, the buyer isn't the user. Long sales cycles and difficult budgetary politics makes it hard to close. Decision makers have different incentives that aren't always clearly aligned with the good of the company as a whole. And so on...